
Introduction
Cleanrooms are highly controlled environments that are meticulously designed to minimize the presence of particles, contaminants, and microorganisms. They are essential for industries where precision, product quality, and sterility are of utmost importance, such as pharmaceuticals, semiconductors, healthcare, electronics manufacturing, aerospace, and more. In order to ensure consistent cleanliness levels and meet industry-specific requirements, International Organization for Standardization (ISO) classifications are utilized as standardized benchmarks. These ISO classifications define the permissible particle counts and airborne microorganism levels within a cleanroom, serving as a crucial tool in cleanroom design, maintenance, and validation. In this post, we will provide a comprehensive overview of cleanroom ISO standards used in different industries.
Understanding ISO Classifications
ISO standards are globally recognized guidelines that define requirements for many industries. The are specific ISO standards for cleanroom environments. These standards play a vital role in ensuring consistency, reliability, and comparability across different industries. Two prominent ISO standards related to cleanrooms are ISO 14644-1 and ISO 14698-1. ISO 14644-1 is measured from Class 1 (cleanest) to Class 9 (least clean) and focuses on airborne particle concentrations. ISO 14698-1 addresses microbiological contamination control, including airborne and surface microbial monitoring and control. Both standards serve as essential references for cleanroom design, validation, and maintenance, facilitating the creation of controlled environments that meet industry-specific needs. Other factors such as temperature and humidity need to be considered, as they can affect particle behavior and microbial growth.
Pharmaceutical Industries
Pharmaceutical operations but abide by both ISO standards and cGMP classifications (graded A trough D). Read more about cGMP cleanroom guidelines here.
- ISO Class 5 (Grade A): The highest level of required cleanliness and is typically found in critical areas, such as aseptic filling suites or biological safety cabinets. It requires a limited number of airborne particles and microorganisms to maintain the sterility of the pharmaceutical products.
- ISO Class 7 (Grade C): Suitable for less critical areas, such as cleanroom packaging or secondary manufacturing areas. It allows for a slightly higher level of airborne particles and microorganisms but still requires controlled conditions.
- ISO Class 8 (Grade D): Applicable to non-sterile manufacturing areas and allows for a higher concentration of airborne particles and microorganisms. These areas are primarily involved in activities such as tablet coating, granulation, or equipment preparation.
Different pharmaceutical processes align with specific ISO classifications. For instance, aseptic filling of injectable medications would typically take place in an ISO Class 5 (Grade A) environment to ensure product sterility. On the other hand, activities like tablet compression or liquid formulation may be carried out in ISO Class 7 (Grade C) or ISO Class 8 (Grade D) environments.
Semiconductor Industry
- ISO Class 1-3 cleanrooms are employed for the most sensitive manufacturing processes. These environments maintain extremely low particle counts and are exceedingly rare (only a few exist in the world), bordering on theoretical levels of cleanliness.
- ISO Class 4-5 cleanrooms are more common and offer slightly higher particle limits compared to Class 1-3. They are still crucial in semiconductor manufacturing, as they help maintain cleanliness during various stages of production. The majority of sensitive operations will take place at this classification.
- ISO Class 6-9 cleanrooms are utilized in less sensitive areas of semiconductor support equipment manufacturing. Although they have higher particle limits, they still maintain controlled environments to prevent contamination and ensure product integrity.
The semiconductor industry faces challenges related to scaling down device sizes, increasing complexity, and evolving technologies. Cleanrooms must address electrostatic discharge (ESD) concerns to prevent damage to sensitive components and manage temperature, humidity, and air filtration systems, which are crucial for meeting these requirements.
Healthcare Industries
- ISO Class 6-7 cleanrooms are commonly employed for critical healthcare processes, such as the production of sterile medications and surgical equipment. These environments have stringent particle count limits, ensuring a low level of contamination.
- ISO Class 8 cleanrooms are utilized for less critical healthcare processes, such as compounding non-sterile medications and manufacturing non-implantable medical devices. Depending on if the medical device is a Class I, Class II or Class III medical device, the cleanroom may need to provide ISO 5 air or ISO 8 air.
- ISO Class 9 cleanrooms can be employed for administrative areas or general storage. While these environments have higher particle limits, they still adhere to cleanliness standards to ensure the overall integrity of healthcare operations.
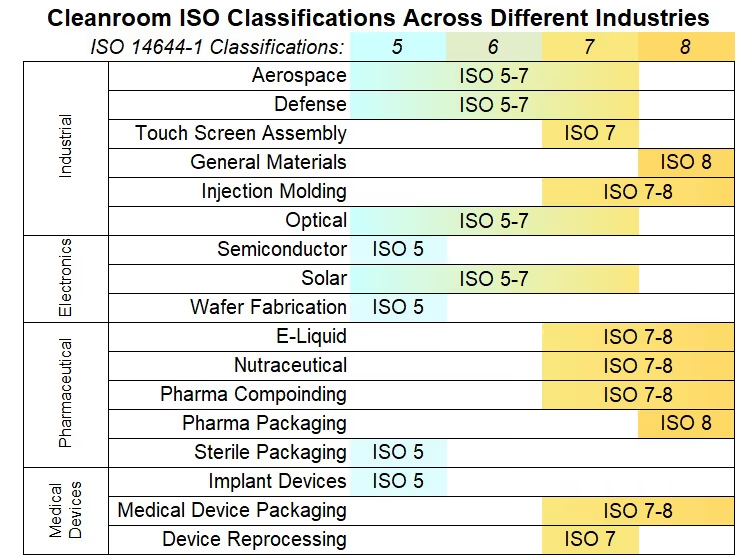
Cleanroom ISO Classifications in Other Industries
- Biotechnology: ISO Class 5 is typically recommended for the production of biologic products like vaccines and therapeutic agents, such as microbial and cell cultures, and biopharmaceuticals. ISO 14698-1 is also important to consider for biotech cleanrooms.
- Electronics: A single 0.3 micron speck of dust could render a failure on a microchip, so ISO Class 5 or 6 is typically recommended for the manufacture of microelectronics and sensitive components that operate on the nanoscale.
- Aerospace and defense: To prevent catastrophic failures, ISO Class 7 or 8 is recommended for manufacture of satellites, spacecraft, and other sensitive, high-tech components. Some more microscale considerations may require cleaner classifications.
- Cell and Gene Therapy: To maintain the high sterility and standardization required for sensitive regenerative medicine treatments, most cell and gene therapy cleanrooms fall within a range from ISO Class 5 to ISO Class 7.
Conclusion
ISO classifications play a crucial role in ensuring cleanroom environments meet industry-specific requirements across various sectors. These standards provide guidelines for airborne particle concentrations and microbiological contamination control, ensuring product quality and safety. Industries such as pharmaceuticals, semiconductors, healthcare, and others rely on ISO classifications to create controlled environments that meet their unique needs. If you are unsure about which ISO classification your operation requires, the experts at Instant Cleanroom Solutions are always happy to help.


